Because not all elements were then known, there were gaps in his periodic table, and Mendeleev successfully used the periodic law to predict properties of some of the missing elements. The first periodic table to become generally accepted was that of the Russian chemist Dmitri Mendeleev in 1869: he formulated the periodic law as a dependence of chemical properties on atomic mass. The underlying reason for these trends is electron configurations of atoms. Trends run through the periodic table, with nonmetallic character (keeping their own electrons) increasing from left to right across a period, and from down to up across a group, and metallic character (surrendering electrons to other atoms) increasing in the opposite direction.
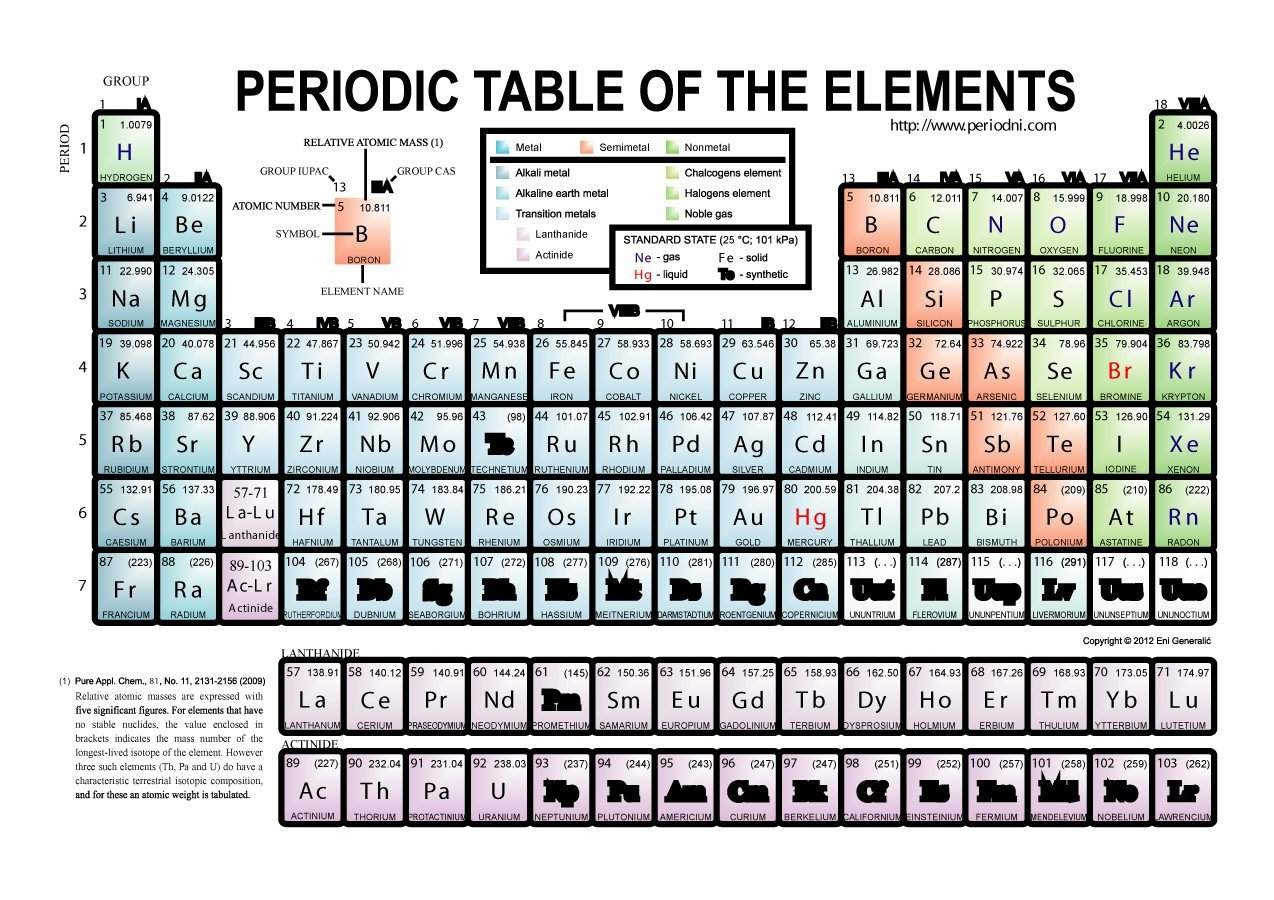
Elements from the same column group of the periodic table show similar chemical characteristics. The rows of the table are called periods, and the columns are called groups. The table is divided into four roughly rectangular areas called blocks. It is a graphic formulation of the periodic law, which states that the properties of the chemical elements exhibit a periodic dependence on their atomic numbers. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of chemistry. We'd like to see a better way of memorising the periodic table - it's even got the four brand new elements that earned a permanent spot in the seventh row back in 2016 (which unfortunately have no cool uses outside of atomic research).Ī version of this article was first published in November 2016.The periodic table, also known as the periodic table of the ( chemical) elements, is a tabular display of the chemical elements.
#Printable periodic table pdf#
You can also download the PDF if you've got a class to teach, or maybe you just want to be great and put it on your bathroom door.Īnd if this whole exercise has made you realise just how rusty you've become with your science basics, check out AsapSCIENCE's Periodic Table Song below. We've included a sneak-peak below, but for the real interactive experience, click here to try it out. I kinda want to tell you all about rubidium and how we use it in the world's most accurate time-keeping devices, and how niobium can help make trains levitate, but you should just check out the periodic table for yourself. The resulting americium is radioactive, and while the tiny amounts of americium dioxide (AmO 2) used in smoke detector produces alpha radiation to sniff out a fire, it will deliver approximately zero radiation to anyone living nearby.
/PeriodicTable-White-58b5d8c15f9b586046df020c.png)
Oh and that very patriotic element, americium? We use that in smoke detectors.įirst unveiled in 1945 during the Manhattan Project, americium is produced by bombarding plutonium with neutrons in a nuclear reactor. You've got strontium for fireworks, and xenon for high-intensity lamps inside lighthouses.

There's thulium for laser eye surgery, cerium for lighter flints, and krypton for flashlights. Lucky for us, Keith Enevoldsen from has come up with this awesome periodic table that gives you at least one example for every single element (except for those weird superheavy elements that don't actually exist in nature).
